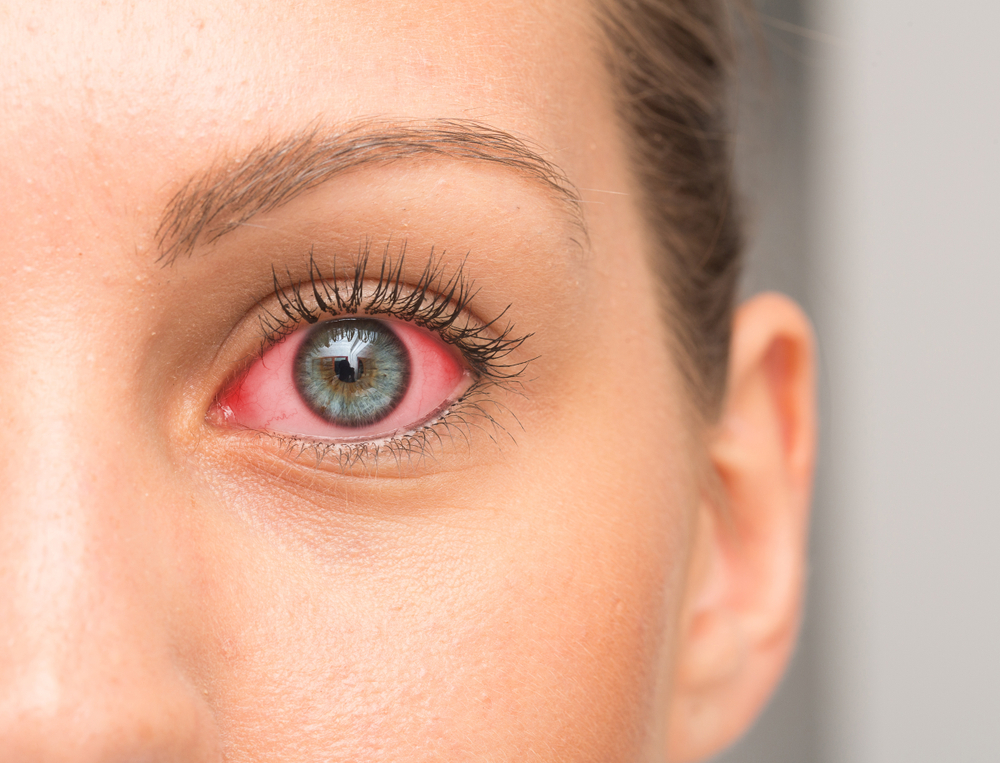
A recent development in eye care may soon provide patients with a more affordable option for treating red eyes. The U.S. Food and Drug Administration (FDA) has tentatively approved a generic version of brimonidine tartrate ophthalmic solution 0.025%, developed by Gland Pharma. This medication is the generic equivalent of the well-known redness relief drop Lumify.
Although the product is not yet available for sale in the United States, this regulatory milestone suggests that patients may soon have access to a lower-cost alternative for managing eye redness.
What Is Brimonidine 0.025%?
Brimonidine tartrate 0.025% is an over-the-counter eye drop designed to relieve redness caused by minor eye irritation. The medication works by selectively constricting certain blood vessels in the eye, which reduces redness while helping avoid the rebound redness often associated with older redness-relief drops.
This lower-dose formulation of brimonidine became widely recognized after Lumify was introduced. Unlike traditional “get-the-red-out” drops that act on multiple receptors, brimonidine is more selective in how it targets blood vessels, which may help reduce the risk of rebound redness when used as directed.
What Does “Tentative Approval” Mean?
When the FDA grants tentative approval, it means the medication has met the agency’s requirements for safety, quality, and effectiveness and has been shown to be bioequivalent to the brand-name drug.
However, the product cannot yet be marketed in the United States because certain regulatory or patent protections for the brand-name product are still in place. Once those exclusivity protections expire, the FDA may grant final approval, allowing the generic medication to be sold.
Why Generic Eye Drops Matter
Generic medications play an important role in improving access to care. Once generic versions become available, they typically lead to:
Lower costs for patients
Greater availability at pharmacies
Increased competition among manufacturers
The U.S. market for brimonidine 0.025% redness-relief drops is estimated to be roughly $39 million annually, demonstrating strong demand for this type of product.
When Should Redness Drops Be Used?
While redness-relieving drops can be helpful for temporary irritation, they should be used appropriately. Occasional redness caused by fatigue, environmental irritation, or mild allergies may respond well to these medications.
However, persistent or recurring red eyes may indicate an underlying condition, such as:
Dry eye disease
Allergic conjunctivitis
Eye infections
Contact lens–related irritation
If redness is accompanied by symptoms such as pain, light sensitivity, discharge, or blurred vision, it is important to schedule an eye examination rather than simply masking the symptom.
The Takeaway
The tentative FDA approval of a generic brimonidine 0.025% eye drop is an encouraging development for patients seeking affordable redness-relief options. Once final approval is granted, generic alternatives to Lumify may soon become available, potentially improving access to treatment for many patients.
As with any eye medication, patients should consult their eye doctor if redness is persistent or if they are unsure about the cause of their symptoms.
Sources
Eyes On Eyecare – “FDA Tentatively Approves Gland Pharma’s Brimonidine Tartrate 0.025% Generic” (March 5, 2026)
FDA Generic Drug Approval Information
Capital Market News – Gland Pharma regulatory update on brimonidine tartrate ophthalmic solution 0.025%









